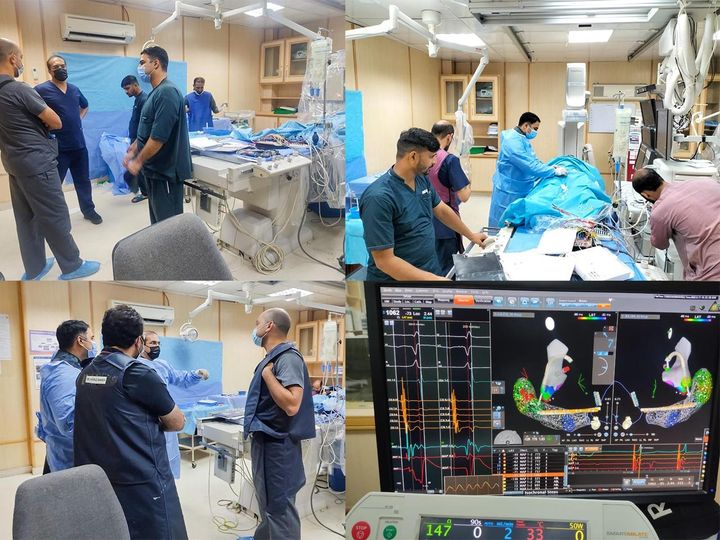
News and updates
Cepheid received FDA clearance for Xpert® BCR-ABL Ultra
Xpert® BCR-ABL Ultra has received clearance from the U.S. Food and Drug Administration for monitoring disease burden in patients with Chronic Myeloid Leukemia (CML). Xpert® BCR-ABL Ultra, an in vitro diagnostic test, is the first FDA-cleared test of its kind capable of delivering accurate molecular results from whole blood samples in under 3 hours.
Xpert® BCR-ABL Ultra is a quantitative test for BCR-ABL major breakpoint (p210) transcripts that provides highly sensitive and on-demand molecular results. Based on the innovative GeneXpert technology, Xpert BCR-ABL Ultra automates the entire test process including RNA isolation, reverse transcription, and fully nested real-time PCR of BCR-ABL target gene and ABL reference gene in one fully automated cartridge.